Radiation
This page is still under construction, using sources and lecture material developed in
these slides: RadiationMadness.pdf
Introduction
Let's play from free word association. I will say a word, and you say the first
word that comes into your mind. Let's give it a try:
- BIRTHDAY
- ICE CREAM
- RADIATION
No doubt, when you got to that last one, a negative word came out of your
mouth. Perhaps something like "danger," or "bad," or "cancer." In popular
culture, "radiation" is associated only with negative outcomes. Yet, few
people understand radiation. Fewer still understand that there are different
kinds - some can cause damage, others cannot.
In this page, we'll explore the definition of radiation, different kinds
of radiation, and finally explore some current topics connected
to the pseudoscientific application of popular knowledge about radiation.
What is "radiation"
Very simply, radiation is the "transport of energy from one point to another without
the need for physical contact between the points." Radiation is everywhere,
and not just in the form you probably think of when someone says "radiation."
Here are different kinds of radiation:
- Sound - sound is a pressure wave (a compression and depression of air molecules)
that travels from the source to the receiver. It is radiation, since
the wave contains energy.
- Light - light is an "electromagnetic" wave, a vibrating electric
and magnetic phenomenon that requires no medium to travel (unlike sound
or water waves). It transports energy.
- Heat - heat is just light that we cannot see because its waves are
too long for our eyes to detector. But we can feel it. The atoms in our
bodies respond to heat and light by vibrating and oscillating, and this
we sense as "heat".
- X-rays - x-rays are just light that has a wavelength so short that
we cannot see it; unlike heat, x-rays can do physical damage
to atoms. This has implication for chemistry, and thus biology.
Radiation can be classified in two groups:
- Ionizing: such radiation has the ability to remove one or more
electrons from an atom. This is called "ionization," and that radiation
is said to be "ionizing." When you remove electrons from an atom,
you alter its chemical properties; it may become highly reactive,
and those reactions can cause changes to nearby molecules. Such processes
are capably of altering a biological system, with potentially
negative consequences that depend on the AMOUNT of ionizing radiation
to which you are exposed. More about that later.
- Non-ionizing: such radiation CANNOT remove electrons from an atom,
and therefore cannot alter their chemical properties. However, this radiation
can still cause atoms inside of molecules to vibrate, can cause the
molecules to spin (rotate), and can excite an electron up into a higher
orbit before the electron then "de-excites" and drops down to its original
energy state. Such radiation has no known negative health effects beyond
heating. More about this later.
Different Examples of Potentially Dangerous Radiation
Ionizing radiation comes in two different forms:
- Particle radiation - so-called "alpha" and "beta" radiation,
discussed below. There are other kinds of particles than can
also ionize, but these are very common and result from the
radioactive decay of naturally occurring elements on Earth.
- Electromagnetic radiation - above a certain frequency,
electromagnetic radiation can ionize. This range is right around
the ultraviolet end of the electromagnetic spectrum.
Any wave with a wavelength shorter than UV can ionize an atom.
"X-rays" and "Gamma rays" are two classes of ionizing electromagnetic
radiation.
Let's discuss these different kinds of ionizing radiation.
- Alpha Particles: these are just the nuclei of helium atoms
(2 protons and 2 neutrons) that have been stripped of their two
orbiting electrons. They have a large electric charge (+2e) and
can ionizing atoms in material by removing electrons from atoms
in the material. However, they are heavy and can be easily stopped
in material. Even a thin sheet of paper can be enough to stop
most alpha radiation! We demonstrate this in class.
- Beta Particles: these are just very fast electrons. They are
often ejected from unstable radioactive nuclei. They have less electric
charge than the alpha particles (-1e) but they are much less massive
and can penetrate further through material before being stopped.
Paper, for instance, is not enough to stop beta particles; but your
hand is enough material to stop them.
- Gamma Radiation: these are electromagnetic waves with wavelengths
much shorter than UV. They carry a lot of energy and EASILY ionize
atoms. They are also very penetrating; because they are just light, which
itself has no mass, they can easily penetrate through a lot of material.
Lead is required to stop them; your hand, or a sheet of paper, are easily
traversed by gamma rays. These, too, result from the decay
of unstable atomic nuclei.
We demonstrate all of the above in class.
How much is too much?
Ionizing radiation doses are measured in "Sieverts," denoted "Sv." A typical US citizen
is exposed to about 6.2 milli-Sieverts each year - half from natural
sources, and half from human sources (e.g. medical diagnostics,
like x-rays). In order to die from exposure to ionizing radiation,
a person must receive 1 Sv ALL AT ONCE - that is almost 200 times
more than a person receives normally in one year.
The following chart is used by the US Nuclear Regulatory Commission (NRC)
in their web page illustrating the typical ionizing radiation dose
in the US, per person, per year. The chart uses units of exposure
called "rems" - 100 milli-rem is equivalent to 1 milli-Sv, so
620 milli-rem is 6.2 milli-Sv.
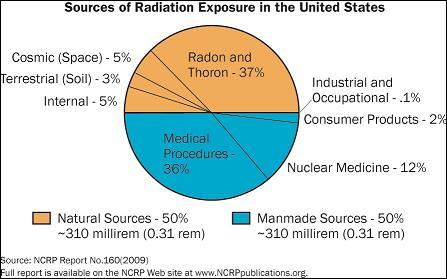
SOURCE: "Fact sheet on Biological Effects of Radiation"
(
http://www.nrc.gov/reading-rm/doc-collections/fact-sheets/bio-effects-radiation.html) and
NCRP Report 160 (2009).
We see that radiation is all around us, and as such our bodies are use to
handling some typical amounts of ionizing radiation, even though
ionizing radiation can in principle be hazardous. This is because
biological systems on Earth evolved under conditions of constant
exposure to such radation - in fact, a biological consequence of
ionizing radiation, called "mutation," is a necessary component
in order to explain the diversity of life on Earth. So ionizing radiation
is, in part, responsible for the vast diversity of life on Earth.
A Demonstration of Ionizing and Non-ionizing Radiation
Atoms are "quantum systems" - that means that you can't just send in
any old electromagnetic wave of any energy and expect to remove
an electron (that is, "ionize" an atom). Atoms must receive a minimum
amount of energy before they will give up their electrons, even
the ones bound furthest from the nucleus. In biological systems,
you have to put in at least as much energy as the weakest chemical
bonds (Hydrogen bonds) in order to disrupt the chemical basis
of the system.
We demonstrate the quantum nature of atoms in class. Please see
this video below for the demonstration.
VIDEO
We learn some very valuable things from this:
- Mobile phone radiation is INCAPABLE of ionizing.
- Green or red laser light are INCAPABLE of ionizing.
- In fact, all the visible light and heat from a set of bright incandescent
bulbs (each about 60 Watts!) is INCAPABLE of ionizing.
- But, the UV-C light from the sanitizing wand,
with a wavelength of about 253 billionths of a meter (nanometers) and a bulb wattage of
just 6 Watts, is easily capable of removing the charge from the aluminum can (ionization!).
The Physics of EM Waves - Energy, Wavelength, and Frequency
Quantum physics, which has withstood repeated testing for about 100 years,
tells us how to relate energy and the properties of EM waves, such
as wavelength and frequency. These properties are illustrated below:
The wavelength and frequency of EM waves are related to
the speed of light, denoted c = 2.998 x 108 m/s:
c = λ * f
where λ is the wavelength (the distance between like parts of neighboring
waves) and f is the frequency of the wave (the number of times per second
that the same part of neighboring waves passes a common point in space). Wavelength
is measured in meters (m) and frequency in a unit called the "Hertz" (Hz) which is
just given by "per second" or 1/s.
Quantum physics also relates the ENERGY carried by the wave to its frequency:
E = h * f
where h is Planck's Constant, h = 4.136 x 10-15 eV*s. An "eV" is
an "electron-Volt," a convenient measure of energy on the scale of an atom.
1 eV is the energy gained by a single electron when it is accelerated
between the two terminals of a 1 Volt battery (for example).
Here is a table illustrating the energy carried by EM waves of different
wavelengths and frequencies:
Let's see if we can get Cas Milner's notes
Let's play word association: I say RADIATION, you say ...
Different kinds
alpha - Apollo mission Pu for thermal generator (movie?)
beta
neutron
gamma, X-ray (chiropractors, mammograms, CT scans) lethal overdose in the news
etc.
Ionizing versus non-ionizing
Demonstration with Geiger-Mueller counter
absorption in materials
1/r^2 law
half-life (dice)
cell phones
*YOU* radiate in the IR
Naturally occurring radiation
cosmic rays - demonstration, has anybody ever flown in a plane? evolution
granite
bananas - B.E.D. salt substitute KCl
brazil nuts
How to dispose of the waste
Yucca Mountain - 1 million years? C'mon!
NIMBY

